 "I'm sure that if they're rejecting things, that's actually what you want to hear," he said. Gortler said he worked at Pfizer for several years, and if it's running behind, it's probably because of its attention to quality. But it slashed vaccine deliveries for some European Union countries this week, according to Reuters. 
Pfizer didn't respond to NPR's request for comment. They described employees working "tirelessly" and volunteering to work Thanksgiving and Christmas. 15, Vice President Pence visited the Catalent contract manufacturing facility making finished vaccine doses for Moderna, where leadership told him they were completing 500,000 doses a day and hoped to double production to meet delivery goals. "Production and releases are not linear and we have explained that we have been successfully scaling up our production yields over time." "We continue to be on track with our expectations of delivering 100 million doses of vaccine by the end of Q1, and 200 million doses by the end of Q2," Moderna spokesperson Ray Jordan wrote in an email to NPR. Moderna said it hasn't been releasing weekly or monthly production estimates, so it couldn't provide more details about how it will deliver significantly more doses to the United States in the coming weeks to meet its first-quarter goal. 
So like I said before, but in this case, just because of the particular nature of this drug and the specific fragility of dealing with anything RNA-related, it's important to take a step back."Īsked about why vaccine delivery appeared to be behind and what's being done to speed it up, Operation Warp Speed spokesperson Michael Pratt in the Trump administration sent NPR the following statement just before the Biden administration took over: "I'd rather hear the companies have fallen short of their production goal, but managed to maintain their quality control because all of this really depends on the quality control," he said. When it comes to COVID-19 vaccines, he said quality is even more important than speed. Building new facilities would require FDA inspections and "take a very long time." He said he understands that Pfizer and Moderna are already working at "maximal capacity" with existing facilities. Gortler is a pharmacist and pharmacologist who specializes in drug quality and supply chain issues. "And RNA is something which is very, very delicate and it can be inactivated, just like - we used to joke - just by looking at it the wrong way." government had agreed to increase the contract for the company’s COVID-19 vaccine by $236 million to roughly $1.25 billion, to include additional costs related to the shot’s studies."Going back to my Yale days when I was a lowly fellow inside of a molecular biology lab, I had to work with RNA myself," he told NPR. 
So far, Johnson & Johnson and AstraZeneca have been the only major global drugmakers with authorized COVID-19 shots that can be stored without a freezer.īoth companies have faced production problems and reports of severe side effects that have slowed uptake of their vaccines. The company would need regulatory sign-off to start shipping vaccine at the higher, refrigerator-level temperatures. 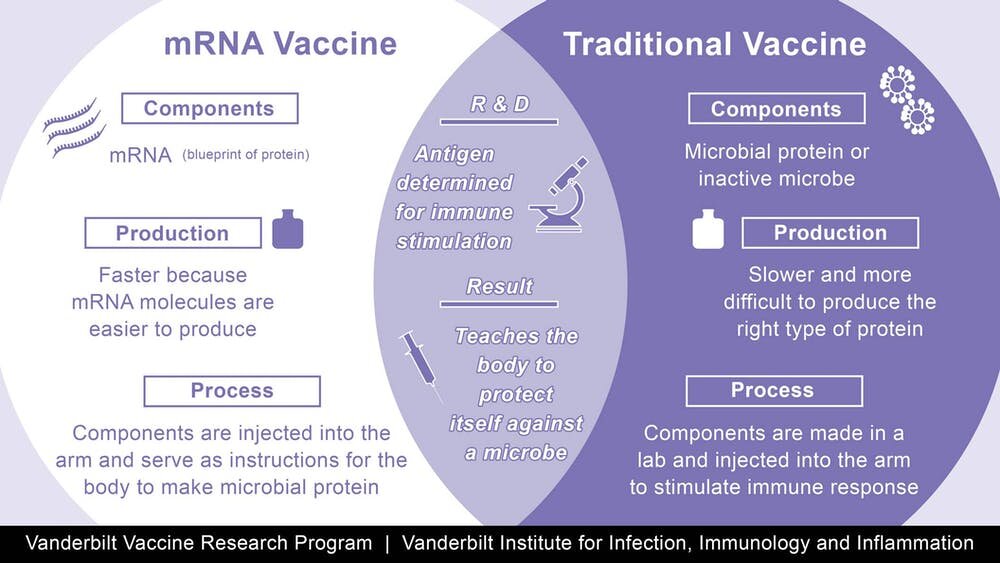
Moderna earlier this month announced shot production deals with Sanofi SA (SASY.PA) and Catalent Inc (CTLT.N). Moderna said it is in advanced talks for additional deals with other manufacturers to help make its shots. drugmaker said it would begin making investments this year and that production boosts would start in late 2021 and carry into early 2022. Moderna's two-dose vaccine uses messenger-RNA technology that programs cells to build immunity to the novel coronavirus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
